Xprecia™
The power of the lab in the palm of your hand
Coagulation PT/INR monitoring
Following on from our successful validation in blood glucose testing, Universal Biosensors (UBI) adapted its electrochemical cell to measure Prothrombin Time and International Normalized Ratio (PT/INR), a test used for monitoring of vitamin K antagonist oral anticoagulation therapy (e.g. Warfarin and Coumadin).
Point-of-care (POC) coagulation testing takes place in three broad settings:
- Hospitals
- Alternative Site Testing, including doctors’ offices, clinics, pharmacies, etc.
- Patients self-testing at home
In 2012, Siemens Healthcare launched the UBI-developed Xprecia Stride™ Coagulation Analyzer and specialized test strips to provide a quick, convenient, and accurate method for medical practitioners to conduct PT/INR testing in the hospital and alternative site testing facilities.
This device is used to monitor the dosage of vitamin K antagonists in patients to ensure its safety and efficacy. Dangerous bleeding events can occur if the ideal dosage of the drug is exceeded; if the dosage is lower than required, the patient is at risk of thrombosis.
The prothrombin time (PT) test allows physicians to appropriately adjust the patient’s dose of the drug to compensate for any diet and lifestyle changes. Globally, approximately 10 million patients are taking warfarin, and over 200 million PT/INR tests are conducted annually to monitor the safe and effective dosage of anticoagulants.
In 2022, UBI introduced the Xprecia Prime™ Coagulation Analyzer, the new and improved coagulation monitoring device for health care professionals. Subsequently, in 2024, UBI launched the Xprecia Prime™ 4U Coagulation Analyzer for patients self-monitoring their PT/INR.
Xprecia Prime™ fits into your palm; is portable, accurate and easy to use. This device is designed for fast and reliable prothrombin time (PT) results displayed in seconds and International Normalised Ratio (INR).
Xprecia Prime™ is approved for sale in the USA through both a 510(k) and CLIA Waiver approval
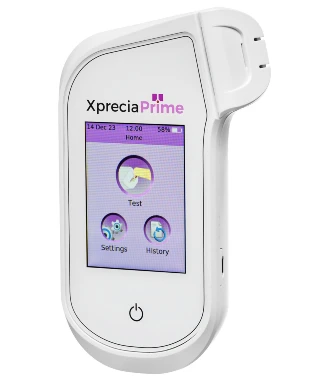
Xprecia Prime™ 4U Coagulation Analyzer

Xprecia Prime™ Coagulation Analyzer
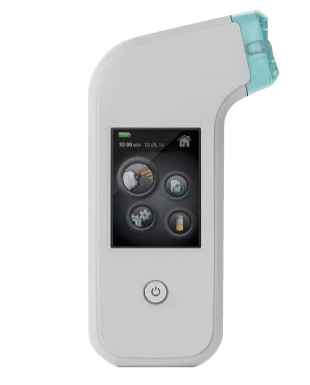
Xprecia Stride™ Coagulation Analyzer
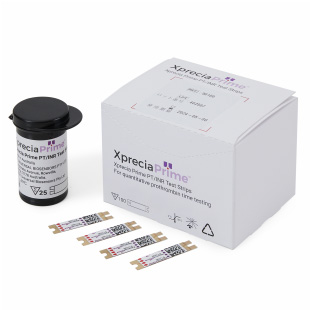
Xprecia Prime™ PT/INR Test Strips

Xprecia Stride™ PT/INR Test Strips
Access to this website is only for healthcare professionals
Please confirm if you are a healthcare professional (HCP) with a special education in healthcare or if you are directly related to the provision of healthcare services.
